Cytoskeletal Dynamics and Actin Sequestering: The TB-500 Research Profile
Delving into the role of the N-acetylated fragment of Thymosin Beta-4 in regulating actin monomer availability. This monograph analyzes how TB-500 influences cell motility and migration patterns within regenerative science models and its hypothesized impact on vascular development.
Jake James Hartley
4/23/20262 min read
The Invisible Architect: How TB-500 Rewires the Cellular Shop Floor
Have you ever stopped to think about how a cell actually moves? It’s not just a blob drifting through the ether. It’s a highly coordinated, energetic process that requires a total re-fit of the cell’s internal architecture. In the world of research, one tiny peptide—TB-500—has become the star of the show for understanding exactly how this structural gymnastics happens.
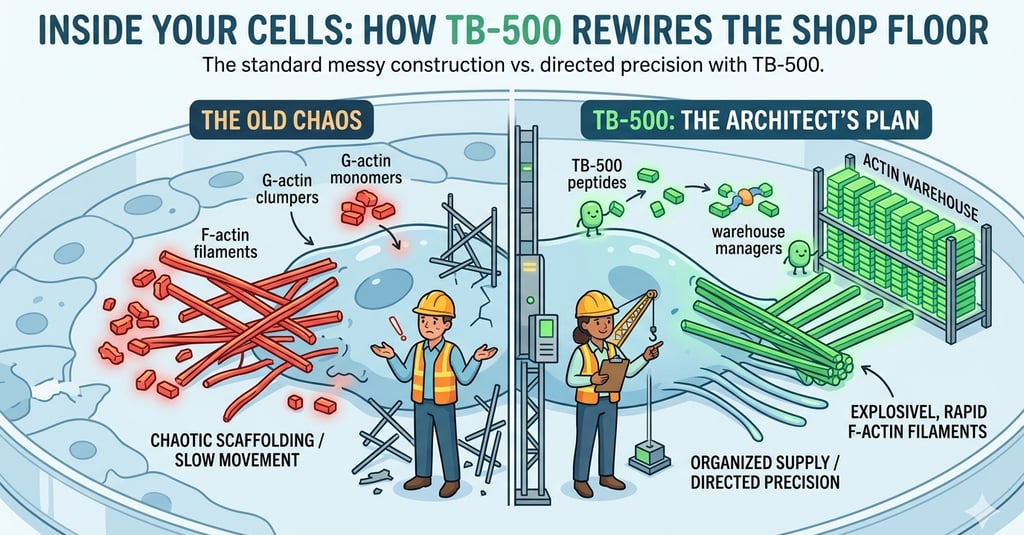
If you’ve ever watched a builder working on a site, you know that progress depends entirely on having the right materials in the right place at the right time. That is exactly what TB-500 does, but on a microscopic scale. To understand how, we need to talk about the cell’s "Lego bricks": actin.
The Great Actin Supply Chain
Inside every cell, there’s a constant tug-of-war between two states of actin. First, you have "G-actin"—these are the loose, free-floating building blocks (the G stands for globular). Then you have "F-actin," which are the long, structural beams (filaments) that give the cell its shape and provide the motor power for movement.
Under normal conditions, actin filaments tend to snap together on their own, a bit like static electricity in a jumper. If a cell left this to chance, its internal scaffolding would be a chaotic mess. It wouldn't be able to move with any purpose.
This is where TB-500 steps in. You can think of it as the most efficient warehouse manager in the business. It binds to the loose G-actin, stopping it from randomly linking up. It holds these "bricks" in reserve, keeping them organised and ready to go. By creating this massive, stable pool of ready-to-use material, the cell isn't reliant on slow, last-minute production. It’s a "just-in-time" supply chain, but for the cell’s skeleton.
Precision Engineering in the "Scratch Test"
So, how do we see this in the lab? Researchers often use something called a "scratch-wound assay." It’s exactly what it sounds like: you take a neat, orderly layer of cells, and you use a tool to make a clean scratch through the middle, leaving a gap.
Then, you watch.
Usually, cells will slowly inch their way across that gap to close it up. It’s a fascinating process of coordination. When you introduce TB-500 to the mix, things get interesting. The cells don't just "try" to move; they move with professional precision.
The TB-500 allows the cells to rapidly dump their reserve of actin at the very front edge of the cell—the "leading edge." This creates the force needed to push the cell forward, creating finger-like protrusions called filopodia that feel out the empty space and pull the cell into the gap. It turns a sluggish, uncoordinated movement into a swift, purposeful crawl.
Why This Matters
This isn't just about cells moving faster; it’s about understanding the fundamental physics of life. By studying TB-500, we’re learning that the secret to rapid structural change isn't always about burning more energy or building more machinery. Sometimes, it’s about better logistics.
TB-500 doesn't do the heavy lifting itself; it simply ensures that the right tools are sitting on the workbench the exact moment the cell decides to move. For researchers peering down the microscope, it’s a brilliant example of how nature manages complex projects—not by brute force, but by clever, highly efficient management of resources. It’s a masterclass in microscopic organisation, and it continues to provide a vital key to understanding how cells organise themselves in the complex, shifting landscape of a living system.
Fit Over 30 +
Contact
Jake@fitover30plus.co.uk
07446899466
© 2024. All rights reserved.


